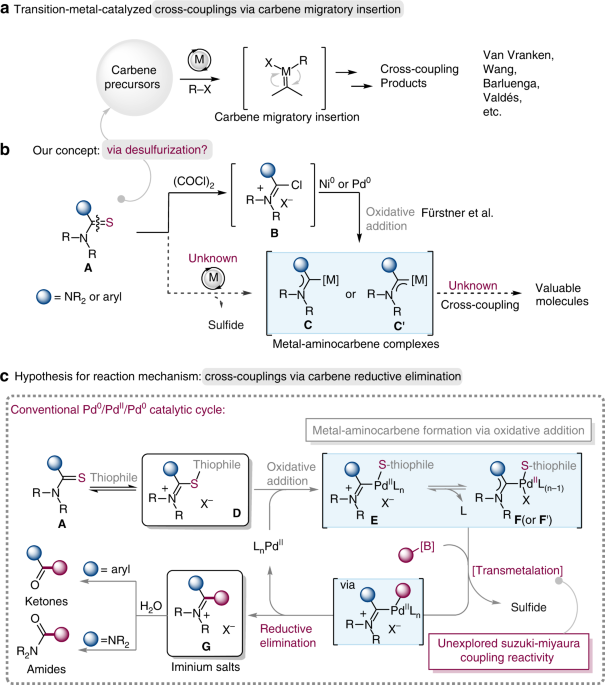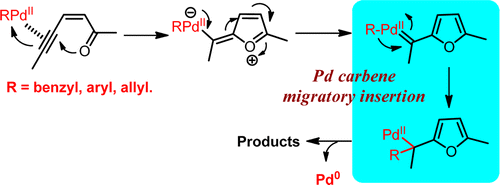
Palladium-catalyzed carbene migratory insertion using conjugated ene-yne-ketones as carbene precursors. - J. Am. Chem. Soc. - X-MOL

PDF) Easy access to medium-sized lactones through metal carbene migratory insertion enabled 1,4-palladium shift

Molecules | Free Full-Text | Site Selectivity in Pd-Catalyzed Reactions of α-Diazo-α-(methoxycarbonyl)acetamides: Effects of Catalysts and Substrate Substitution in the Synthesis of Oxindoles and β-Lactams | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Molecules | Free Full-Text | Site Selectivity in Pd-Catalyzed Reactions of α-Diazo-α-(methoxycarbonyl)acetamides: Effects of Catalysts and Substrate Substitution in the Synthesis of Oxindoles and β-Lactams | HTML
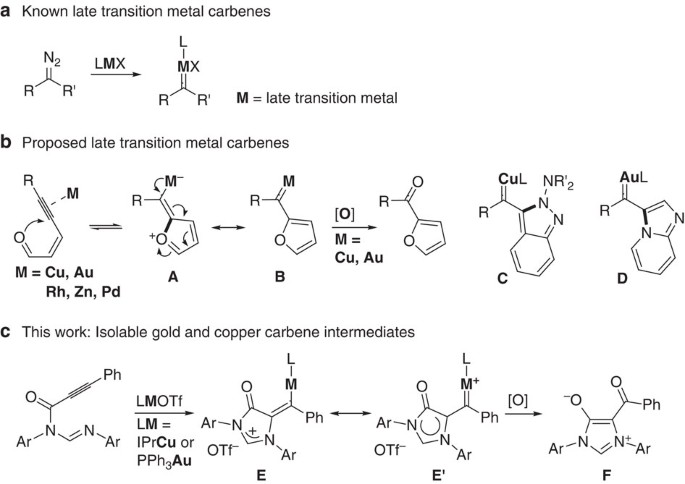
Synthesis and structures of gold and copper carbene intermediates in catalytic amination of alkynes | Nature Communications
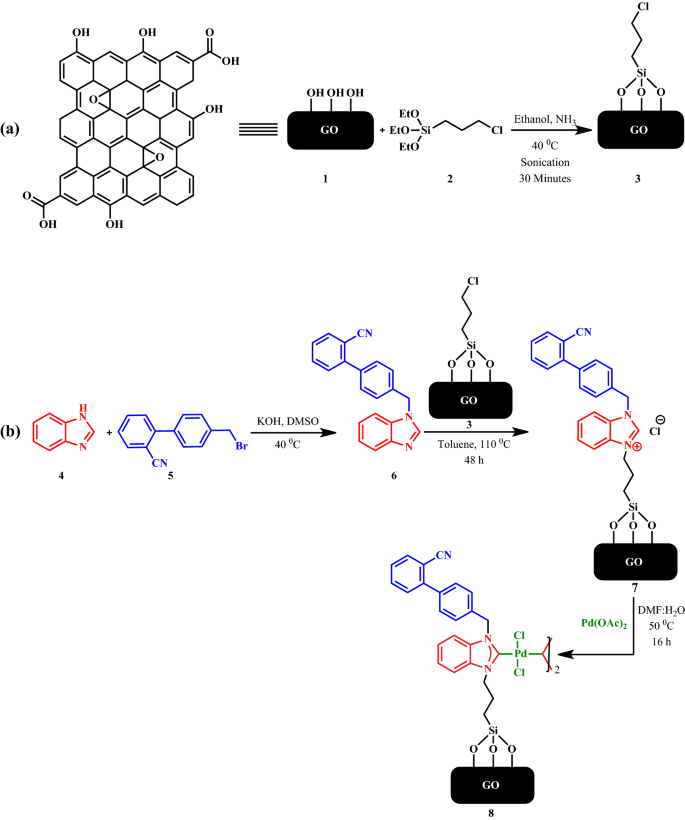
Immobilized N -Heterocyclic Carbene-Palladium(II) Complex on Graphene Oxide as Efficient and Recyclable Catalyst for Suzuki–Miyaura Cross-Coupling and Reduction of Nitroarenes | SpringerLink
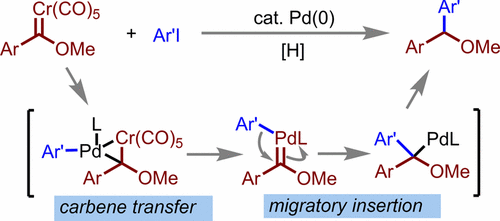
Palladium-Catalyzed Reductive Cross-Coupling Reaction of Aryl Chromium(0) Fischer Carbene Complexes with Aryl Iodides - Organometallics - X-MOL
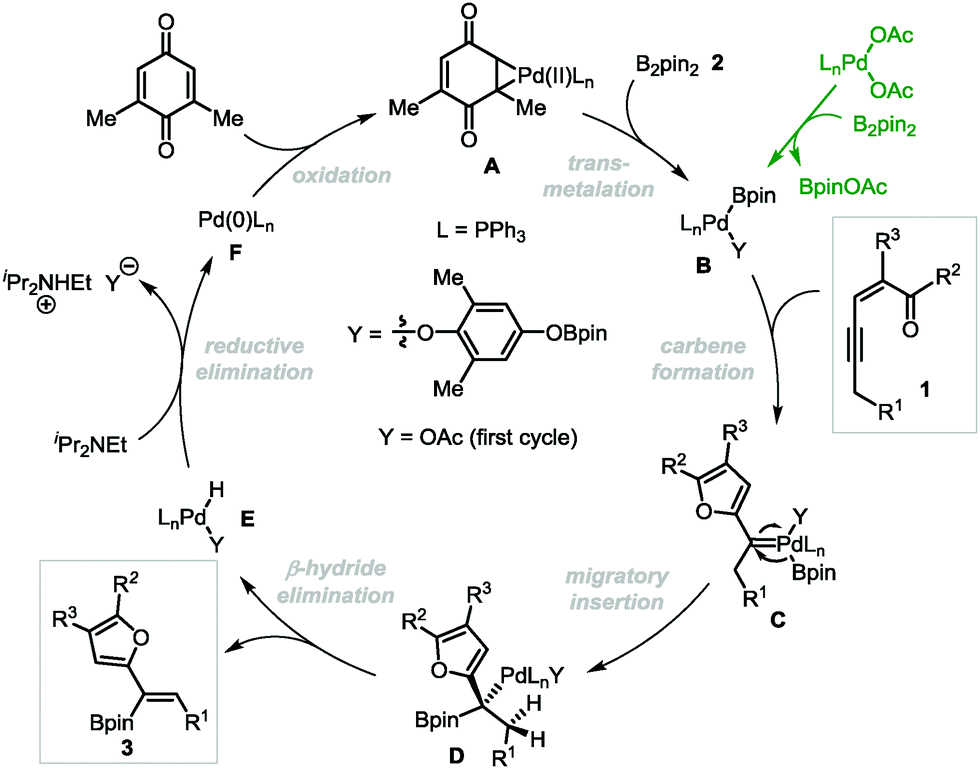
Palladium-catalyzed oxidative borylation of conjugated enynones through carbene migratory insertion: synthesis of furyl-substituted alkenylboronates - Chemical Communications (RSC Publishing)

PDF) Easy access to medium-sized lactones through metal carbene migratory insertion enabled 1,4-palladium shift
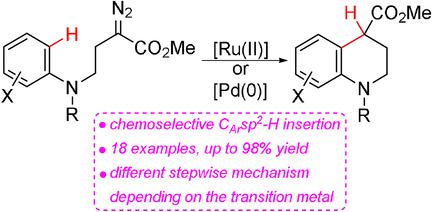
Palladium‐ and Ruthenium‐Catalyzed Intramolecular Carbene CAr−H Functionalization of γ‐Amino‐α‐diazoesters for the Synthesis of Tetrahydroquinolines - Chem. Eur. J. - X-MOL

Reactive Palladium Carbenes: Migratory Insertion and Other Carbene–Hydrocarbyl Coupling Reactions on Well‐Defined Systems - Albéniz - 2018 - European Journal of Inorganic Chemistry - Wiley Online Library
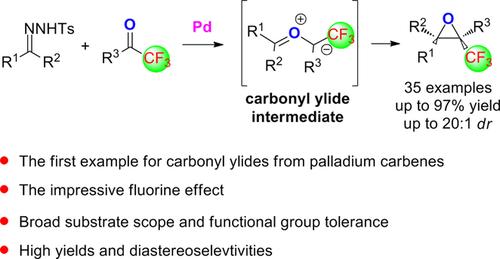
Carbonyl Ylides Derived from Palladium Carbenes: The Impressive Fluorine Effect - Adv. Synth. Catal. - X-MOL

Palladium-catalyzed carbene migratory insertion using conjugated ene-yne-ketones as carbene precursors. | Semantic Scholar

Reactive Palladium Carbenes: Migratory Insertion and Other Carbene–Hydrocarbyl Coupling Reactions on Well‐Defined Systems - Albéniz - 2018 - European Journal of Inorganic Chemistry - Wiley Online Library
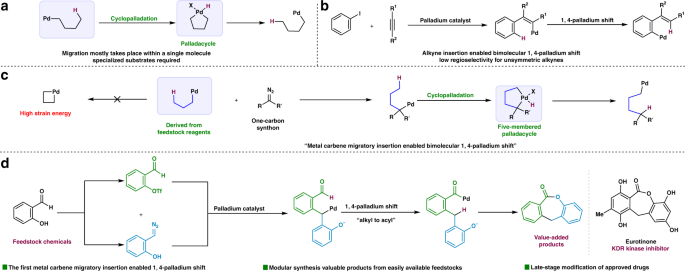
Easy access to medium-sized lactones through metal carbene migratory insertion enabled 1,4-palladium shift | Nature Communications

Reactive Palladium Carbenes: Migratory Insertion and Other Carbene-Hydrocarbyl Coupling Reactions on Well-Defined Systems
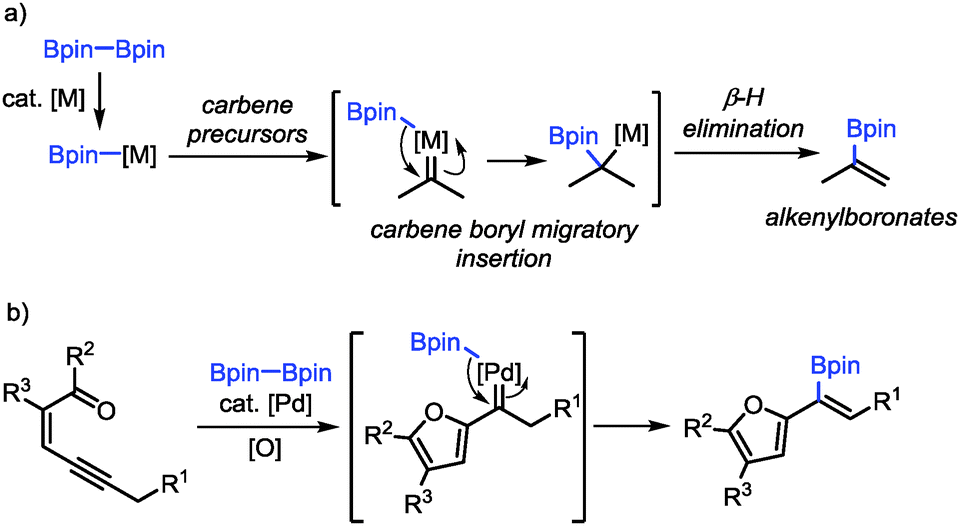
Palladium-catalyzed oxidative borylation of conjugated enynones through carbene migratory insertion: synthesis of furyl-substituted alkenylboronates - Chemical Communications (RSC Publishing)